Dr. Pinaki Ranjan Samanta*, Dr. Manisha, Ms. Ritu Gupta, Ms. Shweta Chauhan and Mr. Prem Kumar Nautiyal, Regulatory Affairs and Analytical Department Uflex Ltd** (Chemical Division), C-5-8, Sector-57, Noida, Uttar Pradesh, India.03.24.22
Editor’s Note: Part 1 appeared in the October 2021 issue of Ink World.
Part 2
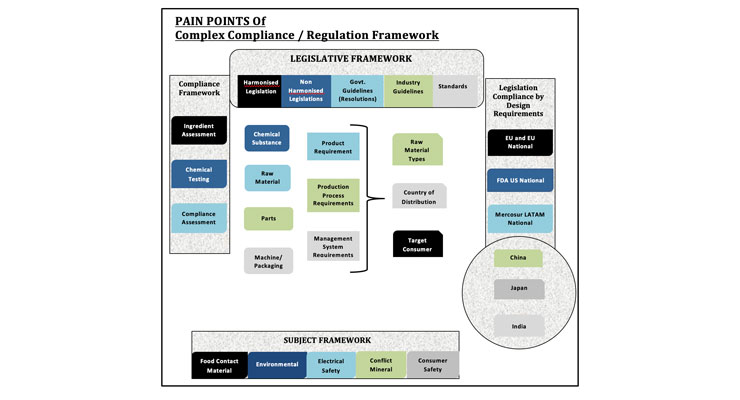
5. REGULATION FRAMEWORK AND PAIN POINTS
Migration tests on consumer goods and packaging including consultancy and evaluation in accordance with Framework Regulation (EC) No. 1935/2004, European Regulation (EU) No. 10/2011 on plastic materials and articles intended to come into contact with foods, European Regulation (EC) No. 282/2008 on recycled plastic materials and articles intended to come into contact with foods, and Regulation (EC) No. 450/2009 on active and intelligent packaging. Most countries had its own relevant legislation along with local requirements, some are as follows:
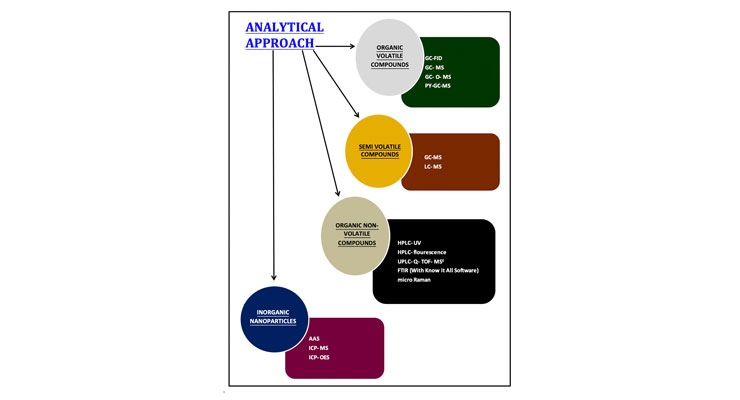
6. ANALYTICAL APPROACH: NIAS TESTING AND RISK ASSESSMENT SOLUTIONS
Several guidance documents have been published which offer some clues on how to conduct analytical assessment of NIAS in food contact materials and articles. However, the recommendation of these guidance documents are not legally binding but scientists must follow the steps given below to analyze NIAS components:
Accumulating Information and Data Collection: Scientists and regulatory experts gather information regarding the chemical composition of the food contact material to achieve a complete (as far as possible) understanding of the material composition, including all relevant information from the manufacturing process along with traceability of raw materials supplier’s data.
Process Validation and Analytical Screenings: Analytical teams conduct investigations using a series of analytical approaches to attempt to determine all potential migrants. NIAS screening programs are executed with a wide range of sample preparation techniques (e.g. extraction with solvents under controlled conditions) and analytical techniques (thermal desorption gas chromatography time-of flight mass spectrometry (GC/Q-TOF) gas chromatography mass spectrometry (GC-MS), liquid chromatography – mass spectroscopy (LC-MS), inductively coupled plasma mass spectrometry (ICP-MS)) to identify and semi-quantify the presence of NIAS and heavy metals, inorganics etc.
Identification and Estimate of Hazardous Substances: Toxicology professionals identify the presence of any hazardous substances based on literature surveys and/or based on internationally recognized scientific principles on risk assessment (e.g. the Threshold of Toxicological Concern-TTC approach /Cramer classification).
Exposure Assessment: Scientists and regulatory experts’ teams perform migration studies and/or exposure assessments in order to define real exposure and estimated daily intake.
Final Risk Assessment: Regulatory and toxicology expert to calculate the risk and hazard to the consumer and make any recommendations if required. Make a documented report on any potential health risk associated with the final material or article.
Heavy metals from FCM can be analyzed by ICP-MS after leaching of packaging materials with 10% dilute hydrochloric acid. Migration of organic compounds from packaging materials debris are commonly carried out using different food simulants: liquid (e.g., 95% ethanol, 3% acetic acid), and/or solid powder (Tenax®). Analytical methods applied for the analysis of food simulants depend on their chemical properties. Examples of analytical methods for screening of volatile and non-volatile migrants in different food simulants can be seen in following Table 1.
Naturally, if target analysis is performed, other analytical methods such as gas chromatography coupled to flame ionization detector (GC-FID), gas chromatography coupled to mass spectrometry with pyrolyzer (Py-GC-MS), high-performance liquid chromatography coupled to an ultraviolet detector (HPLC-UV) and high-performance liquid chromatography coupled to a fluorescence detector (HPLC-fluorescence), and ultra-high-performance liquid chromatography coupled to the triple quadrupole mass spectrometry (UPLC-MS-QQQ)—can be applied with automatic detection capabilities and filtering procedures. MS and tandem mass spectrometry (MS/MS) data to be processed to find chemical formulae and structure along with toxicological information for the most abundant analytes in FCMs.
The research on the potential toxic compounds from plastic materials, printing inks, laminating adhesives for food contact applications are still undertaken and the law is constantly
being improved.
Non-volatile compounds are determined by high performance liquid chromatography (HPLC). Here the identification of substances is more difficult as no extensive mass spectra databases are available.
The reason for this is that different ionisation modes can be applied depending on the different properties of the analytes, which leads to different fragmentation patterns.
Furthermore, the setting of the analytical instrument and the conditions in the ion source also influence the fragmentation pattern, which makes a comparison of obtained mass spectra between different laboratories difficult.
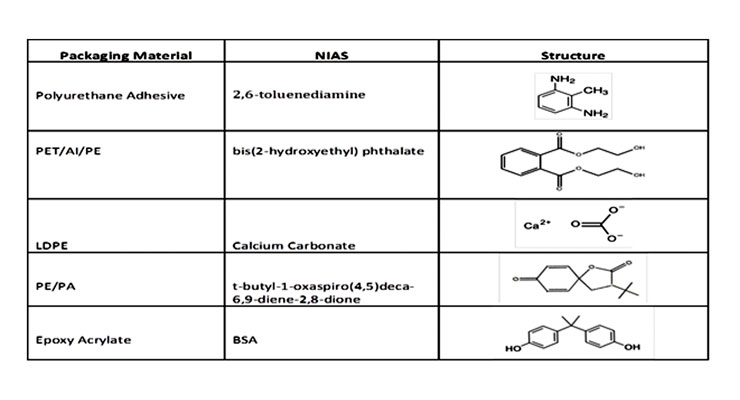
Primary aromatic amines (PAAs) which can occur in colored or printed materials as residual educts from the synthesis of azo pigments or as degradation products of azo pigments generated by reductive cleavage.
PAAs can also occur as reaction products of isocyanates formed by degradation of polyurethane adhesives. Such an LC-MS/MS multi-analyte method focussing on PAAs deriving from azo pigments and isocyanates typically used in food contact materials is currently being developed by different commercial and research laboratories.
7. GAPS and HURDLES
The fact that the identity of NIAS and NLS are usually not or incompletely known to the manufacturer of a food contact material (FCM) as well as the required detection limit of < 10 μg/kg food (simulant) pose a challenge to their analytical determination. For unknown substances, non-target screening tests need to be applied.
The analytical method of choice should be able to detect, identify and quantify a wide variety of substances differing in their physical and chemical properties.
The mode of detection should be independent from the detected molecule, i.e. the response of the selected detector should be the same or similar in order to enable a (semi-)quantification of the detected substances. NIAS with unknown toxicity, but known structure, the threshold of toxicological concern (TTC) can be applied. It is a screening tool for qualitative risk assessment. Scientists face the following hurdles during analysis and data accumulations:
a. Lack of common grounds on materials regulated by safety data with multiple languages and a magnitude of national rules.
b. Variations in types of restriction.
c. Commonality between national legislation and industry guidelines.
d. Multiple national requirements and mutual recognition or self-regulation.
e. Burden for multiple testing and method development.
f. Higher expertise scientist needed for analysis.
g. Afraid of legal cases or financial debit note due to the failure of mutual recognition.
h. Global market for non- harmonized materials.
8. CONCLUSION
It must be noted that no single analytical method is able to detect all possible migratable NIAS. Different screening approaches need to be applied for different groups of substances.
A combination of several screening approaches will yield the maximum of information. The more is known about IAS, the better the chances to identify NIAS because then the screening methods applied can be adapted to the physical and chemical properties of the groups of substances expected.
The analysis of food is a very difficult task due to the complexity of the matrix.
Acknowledgement
The authors are overwhelmed in all humbleness and gratefulness to acknowledge for written this article to Uflex management who helped to put these ideas well above the level of simplicity and something concrete.
We are thankful to Uflex Ltd - Chemical Division for providing infrastructure and constructive criticism which have helped ours to accomplish this work. It is a genuine pleasure to express our deep sense of thanks and gratitude to our colleagues for their timely advice, meticulous scrutiny, scholastic advice and scientific approach to a very great extent.
Authors
Dr. Pinaki Ranjan Samanta has more than 26 years of experience in the areas associated to industrial research of coating and testing of finished products and raw materials of coating-related products. He received M. SC and Ph. D in the field of organic chemistry, also done PG diploma in paint technology.
Dr. Manisha, masters in chemistry from IIT and Ph.D in chemistry with more than nine years of experience in the field of analytical and polymer analysis is currently with Uflex Limited, Noida as manager (R&D).
Ms. Ritu Gupta, masters in chemistry, is currently working with Uflex Limited, Noida as chemist (R&D instrumentation). She has research experience of more than two years with the expertise in the area of analysis by using state-of-the-art instruments like Py- GC-MS, GC-HS, FTIR, UV-spectrophotometer etc.
Ms. Shweta Chauhan is an engineering graduate and has done B.Tech.in chemical engineering. She is a regulatory expert and currently working with Uflex Limited, Chemical Division, Noida.
Mr. Prem Kumar Nautiyal, a post graduate and M. Phil in chemistry along with certified black belt and PGDMM with 32 years of industrial experience in various industries and he is a hardcore synthetic chemist with specialty in design and development of polyurethane laminating adhesive. He is currently working with Uflex Limited (Chemical Division) as HOD - R & D, accountable for development of new formulations and products.
Reference
1. Printing ink, polymers and food packaging – Insight into the unique challenge and vulnerability towards compliance of materials and global regulatory issues - Part 1 and Part 2; by Dr. Pinaki Ranjan Samanta, Dr. Ruchi Gupta, Ms. Ritu Gupta, Ms. Shweta Chauhan and Mr. Anuj Johri ; April –May 2020: www.polymerspaintcolourjournal.com/issues or www.coatingsgroup.com.
2. Analytical Approaches for Analysis of Safety ofModern Food Packaging: A Review; by MagdalenaWrona and Cristina Nerín;Department of Analytical Chemistry, Aragon Institute of Engineering Research I3A, University of Zaragoza,María de Luna, 3, 50018 Zaragoza, Spain; Molecules 2020, 25, 752; doi:10.3390/molecules25030752; www.mdpi.com/journal/molecules.
3. Review of analytical approaches for the identification of non-intentionally added substances in paper and board food contact materials, by Ruud J.B. Peters, Iris Groeneveld, Patricia Lopez Sanchez, WouterGebbink, ArjenGersen, Monique de Nijs, Stefan P.J. van Leeuwen; Trends in Food Science & Technology,https://doi.org/10.1016/j.tifs.2018.12.010.
4. Articles from Packaginglaw.com; A resource provide by Kehller and Heckman LLP.
5. EuPIA Guidance on Migration Test Methods for the evaluation of substances in printing inks and varnishes for food contact materials ; 2017-07-31; European Printing Ink Association · EuPIA - a sector of CEPE aisbl; www.eupia.org.
6. Dutra, C.; Pezo, D.; de AlvarengaFreire, M.T.; Nerín, C.; Reyes, F.G.R. Determination of volatileorganiccompounds in recycled polyethylene terephthalate and high-density polyethylene by headspace solid phasemicroextraction gas chromatography mass spectrometry to evaluate the e_ciency of recycling processes.J. Chromatogr. A 2011, 1218, 1319–1330.
7. Analytics of Substances in Food Packaging Material: C. Gehring https://analyticalscience.wiley.com/do/ 10.1002/gitlab.16086.
Disclaimer: The views expressed are solely the corresponding author’s personal opinion. Uflex Ltd will not accept any liability for any loss or damage that may occur from the use of this information nor do we offer a warranty against patent infringement.
Part 2
5. REGULATION FRAMEWORK AND PAIN POINTS
Migration tests on consumer goods and packaging including consultancy and evaluation in accordance with Framework Regulation (EC) No. 1935/2004, European Regulation (EU) No. 10/2011 on plastic materials and articles intended to come into contact with foods, European Regulation (EC) No. 282/2008 on recycled plastic materials and articles intended to come into contact with foods, and Regulation (EC) No. 450/2009 on active and intelligent packaging. Most countries had its own relevant legislation along with local requirements, some are as follows:
6. ANALYTICAL APPROACH: NIAS TESTING AND RISK ASSESSMENT SOLUTIONS
Several guidance documents have been published which offer some clues on how to conduct analytical assessment of NIAS in food contact materials and articles. However, the recommendation of these guidance documents are not legally binding but scientists must follow the steps given below to analyze NIAS components:
Accumulating Information and Data Collection: Scientists and regulatory experts gather information regarding the chemical composition of the food contact material to achieve a complete (as far as possible) understanding of the material composition, including all relevant information from the manufacturing process along with traceability of raw materials supplier’s data.
Process Validation and Analytical Screenings: Analytical teams conduct investigations using a series of analytical approaches to attempt to determine all potential migrants. NIAS screening programs are executed with a wide range of sample preparation techniques (e.g. extraction with solvents under controlled conditions) and analytical techniques (thermal desorption gas chromatography time-of flight mass spectrometry (GC/Q-TOF) gas chromatography mass spectrometry (GC-MS), liquid chromatography – mass spectroscopy (LC-MS), inductively coupled plasma mass spectrometry (ICP-MS)) to identify and semi-quantify the presence of NIAS and heavy metals, inorganics etc.
Identification and Estimate of Hazardous Substances: Toxicology professionals identify the presence of any hazardous substances based on literature surveys and/or based on internationally recognized scientific principles on risk assessment (e.g. the Threshold of Toxicological Concern-TTC approach /Cramer classification).
Exposure Assessment: Scientists and regulatory experts’ teams perform migration studies and/or exposure assessments in order to define real exposure and estimated daily intake.
Final Risk Assessment: Regulatory and toxicology expert to calculate the risk and hazard to the consumer and make any recommendations if required. Make a documented report on any potential health risk associated with the final material or article.
Heavy metals from FCM can be analyzed by ICP-MS after leaching of packaging materials with 10% dilute hydrochloric acid. Migration of organic compounds from packaging materials debris are commonly carried out using different food simulants: liquid (e.g., 95% ethanol, 3% acetic acid), and/or solid powder (Tenax®). Analytical methods applied for the analysis of food simulants depend on their chemical properties. Examples of analytical methods for screening of volatile and non-volatile migrants in different food simulants can be seen in following Table 1.
Naturally, if target analysis is performed, other analytical methods such as gas chromatography coupled to flame ionization detector (GC-FID), gas chromatography coupled to mass spectrometry with pyrolyzer (Py-GC-MS), high-performance liquid chromatography coupled to an ultraviolet detector (HPLC-UV) and high-performance liquid chromatography coupled to a fluorescence detector (HPLC-fluorescence), and ultra-high-performance liquid chromatography coupled to the triple quadrupole mass spectrometry (UPLC-MS-QQQ)—can be applied with automatic detection capabilities and filtering procedures. MS and tandem mass spectrometry (MS/MS) data to be processed to find chemical formulae and structure along with toxicological information for the most abundant analytes in FCMs.
The research on the potential toxic compounds from plastic materials, printing inks, laminating adhesives for food contact applications are still undertaken and the law is constantly
being improved.
Non-volatile compounds are determined by high performance liquid chromatography (HPLC). Here the identification of substances is more difficult as no extensive mass spectra databases are available.
The reason for this is that different ionisation modes can be applied depending on the different properties of the analytes, which leads to different fragmentation patterns.
Furthermore, the setting of the analytical instrument and the conditions in the ion source also influence the fragmentation pattern, which makes a comparison of obtained mass spectra between different laboratories difficult.
Primary aromatic amines (PAAs) which can occur in colored or printed materials as residual educts from the synthesis of azo pigments or as degradation products of azo pigments generated by reductive cleavage.
PAAs can also occur as reaction products of isocyanates formed by degradation of polyurethane adhesives. Such an LC-MS/MS multi-analyte method focussing on PAAs deriving from azo pigments and isocyanates typically used in food contact materials is currently being developed by different commercial and research laboratories.
7. GAPS and HURDLES
The fact that the identity of NIAS and NLS are usually not or incompletely known to the manufacturer of a food contact material (FCM) as well as the required detection limit of < 10 μg/kg food (simulant) pose a challenge to their analytical determination. For unknown substances, non-target screening tests need to be applied.
The analytical method of choice should be able to detect, identify and quantify a wide variety of substances differing in their physical and chemical properties.
The mode of detection should be independent from the detected molecule, i.e. the response of the selected detector should be the same or similar in order to enable a (semi-)quantification of the detected substances. NIAS with unknown toxicity, but known structure, the threshold of toxicological concern (TTC) can be applied. It is a screening tool for qualitative risk assessment. Scientists face the following hurdles during analysis and data accumulations:
a. Lack of common grounds on materials regulated by safety data with multiple languages and a magnitude of national rules.
b. Variations in types of restriction.
c. Commonality between national legislation and industry guidelines.
d. Multiple national requirements and mutual recognition or self-regulation.
e. Burden for multiple testing and method development.
f. Higher expertise scientist needed for analysis.
g. Afraid of legal cases or financial debit note due to the failure of mutual recognition.
h. Global market for non- harmonized materials.
8. CONCLUSION
It must be noted that no single analytical method is able to detect all possible migratable NIAS. Different screening approaches need to be applied for different groups of substances.
A combination of several screening approaches will yield the maximum of information. The more is known about IAS, the better the chances to identify NIAS because then the screening methods applied can be adapted to the physical and chemical properties of the groups of substances expected.
The analysis of food is a very difficult task due to the complexity of the matrix.
Acknowledgement
The authors are overwhelmed in all humbleness and gratefulness to acknowledge for written this article to Uflex management who helped to put these ideas well above the level of simplicity and something concrete.
We are thankful to Uflex Ltd - Chemical Division for providing infrastructure and constructive criticism which have helped ours to accomplish this work. It is a genuine pleasure to express our deep sense of thanks and gratitude to our colleagues for their timely advice, meticulous scrutiny, scholastic advice and scientific approach to a very great extent.
Authors
Dr. Pinaki Ranjan Samanta has more than 26 years of experience in the areas associated to industrial research of coating and testing of finished products and raw materials of coating-related products. He received M. SC and Ph. D in the field of organic chemistry, also done PG diploma in paint technology.
Dr. Manisha, masters in chemistry from IIT and Ph.D in chemistry with more than nine years of experience in the field of analytical and polymer analysis is currently with Uflex Limited, Noida as manager (R&D).
Ms. Ritu Gupta, masters in chemistry, is currently working with Uflex Limited, Noida as chemist (R&D instrumentation). She has research experience of more than two years with the expertise in the area of analysis by using state-of-the-art instruments like Py- GC-MS, GC-HS, FTIR, UV-spectrophotometer etc.
Ms. Shweta Chauhan is an engineering graduate and has done B.Tech.in chemical engineering. She is a regulatory expert and currently working with Uflex Limited, Chemical Division, Noida.
Mr. Prem Kumar Nautiyal, a post graduate and M. Phil in chemistry along with certified black belt and PGDMM with 32 years of industrial experience in various industries and he is a hardcore synthetic chemist with specialty in design and development of polyurethane laminating adhesive. He is currently working with Uflex Limited (Chemical Division) as HOD - R & D, accountable for development of new formulations and products.
Reference
1. Printing ink, polymers and food packaging – Insight into the unique challenge and vulnerability towards compliance of materials and global regulatory issues - Part 1 and Part 2; by Dr. Pinaki Ranjan Samanta, Dr. Ruchi Gupta, Ms. Ritu Gupta, Ms. Shweta Chauhan and Mr. Anuj Johri ; April –May 2020: www.polymerspaintcolourjournal.com/issues or www.coatingsgroup.com.
2. Analytical Approaches for Analysis of Safety ofModern Food Packaging: A Review; by MagdalenaWrona and Cristina Nerín;Department of Analytical Chemistry, Aragon Institute of Engineering Research I3A, University of Zaragoza,María de Luna, 3, 50018 Zaragoza, Spain; Molecules 2020, 25, 752; doi:10.3390/molecules25030752; www.mdpi.com/journal/molecules.
3. Review of analytical approaches for the identification of non-intentionally added substances in paper and board food contact materials, by Ruud J.B. Peters, Iris Groeneveld, Patricia Lopez Sanchez, WouterGebbink, ArjenGersen, Monique de Nijs, Stefan P.J. van Leeuwen; Trends in Food Science & Technology,https://doi.org/10.1016/j.tifs.2018.12.010.
4. Articles from Packaginglaw.com; A resource provide by Kehller and Heckman LLP.
5. EuPIA Guidance on Migration Test Methods for the evaluation of substances in printing inks and varnishes for food contact materials ; 2017-07-31; European Printing Ink Association · EuPIA - a sector of CEPE aisbl; www.eupia.org.
6. Dutra, C.; Pezo, D.; de AlvarengaFreire, M.T.; Nerín, C.; Reyes, F.G.R. Determination of volatileorganiccompounds in recycled polyethylene terephthalate and high-density polyethylene by headspace solid phasemicroextraction gas chromatography mass spectrometry to evaluate the e_ciency of recycling processes.J. Chromatogr. A 2011, 1218, 1319–1330.
7. Analytics of Substances in Food Packaging Material: C. Gehring https://analyticalscience.wiley.com/do/ 10.1002/gitlab.16086.
Disclaimer: The views expressed are solely the corresponding author’s personal opinion. Uflex Ltd will not accept any liability for any loss or damage that may occur from the use of this information nor do we offer a warranty against patent infringement.