Sneha Bhatia, George Fuchs and Gregory Pace03.15.19
Overview
Printing inks are formulations of mixtures made from “raw materials,” which are comprised of chemical substances. A common problem in chemical hazard classification of mixtures is assigning the appropriate chemical classification and resolving discrepancies in classification.
It is often found that the same printing ink chemicals may be classified differently by different suppliers. The discrepancies in chemical classification stem partially from differences in classification of chemicals across various countries, data availability, interpretation of data and different concentration cut-offs.
As such, users of the raw material are often left with the task of assessing the data and resolving the classification discrepancies.
This is a crucial issue, especially for chemicals that may be classified as CMR (carcinogens, mutagens or reproductive toxins). To provide clarity and eliminate confusion in the customer base (i.e. printers), the NAPIM SDS Evaluation Committee, composed of EHS experts from both ink manufacturers and raw materials suppliers, has undertaken a program aimed at identifying printing ink raw materials which are reported to have health hazard classifications that are inconsistent or conflict with the health hazard classifications of similar/identical substances.
Introduction
Classification of a chemical or a mixture is a scientific assessment of whether it has the potential to harm human health or the environment if exposure occurs. Chemicals are evaluated and classified so that people using them can understand any potential hazardous effects they could have on human health or the environment and protect against that harm.
It is vital that classification is based on accurate, robust and adequate data. The purpose of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is to protect human health and the environment by having readily available information on the hazard properties of chemicals.
This permits the development of appropriate control measures that allow the production, transport, use and disposal of chemicals to be managed safely. The Safety Data Sheet (SDS) identifies the hazards of chemicals and mixtures and communicates the hazard information, as well as protective measures, on labels and SDS.
In 2015, the Occupational Safety and Health Administration (OSHA) integrated the GHS ver. 3 into the Hazard Communication Standard (HCS) 29CFR 1910.1200. This revised federal regulation was to be implemented in the US by June 1, 2015. The revised rule included new pictograms, signal word, hazard statements and precautionary statements appearing on the labels and SDS.
GHS is revised frequently. As of 2018, the most current GHS is version VII. OSHA adopted GHS ver. 3. The OSHA HazCom standard does not change automatically to when the GHS is revised. Classification of chemicals using the OSHA HazCom standard under Appendices A and B may not be consistent with the version of GHS in use in other geographic regions.
Accordingly, to provide clarity and eliminate confusion for employees and customers, the NAPIM SDS Evaluation committee has undertaken a program aimed at identifying printing ink raw materials which are known to have health hazard classifications that are inconsistent with the health hazard classification procedure specified in Appendix A of the HazCom standard. These materials along with their recommended health hazard classification are listed below. This applies to printing inks sold in the United States only.
Process
The NAPIM SDS Evaluation Committee, composed of experts within the NAPIM membership, has undertaken a program aimed at identifying printing ink materials which are known to have health hazard classifications that are inconsistent with the health hazard classification procedure specified in Appendix A of the HazCom standard. The printing ink materials with discrepancies in hazard classifications are nominated to the NAPIM SDS Evaluation committee.
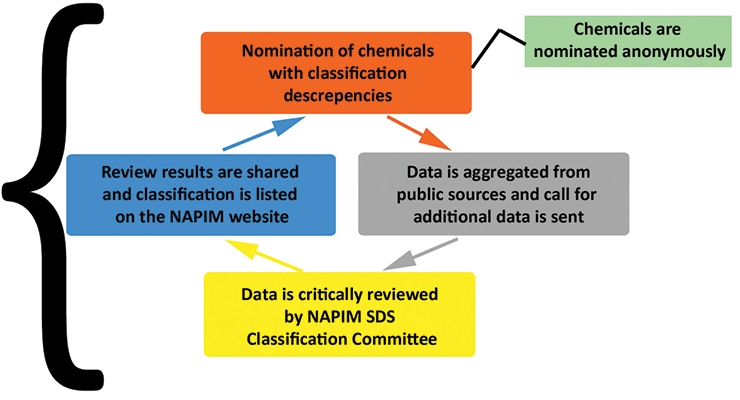
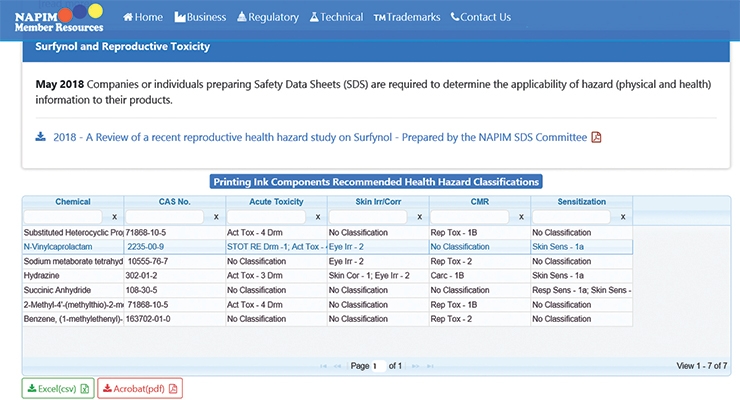
This is an anonymous process. The committee reviews all available health hazard data (see Figure 1) to derive a consensus recommendation that is based on the available data. When a consensus health hazard classification is reached, the material along with its recommended health hazard classification is listed on the NAPIM website (see Figure 2). This is applicable to the printing inks sold in the US only.
Conclusion
The NAPIM SDS Committee process for resolving health hazard classification discrepancies utilizes collective knowledge and ensures transparency in resolving the health hazard classification discrepancies. The printing ink industry is committed to the safety of its employees and customers. Providing consistent and accurate information regarding potential printing ink health hazards to both employees and customers is essential to meeting this commitment.
Sneha Bhatia is a regulatory affairs specialist of global regulatory product stewardship at Sun Chemical. George Fuchs is the director of regulatory affairs and technology at NAPIM. Gregory Pace is the manager of global regulatory & product stewardship at Sun Chemical.
Printing inks are formulations of mixtures made from “raw materials,” which are comprised of chemical substances. A common problem in chemical hazard classification of mixtures is assigning the appropriate chemical classification and resolving discrepancies in classification.
It is often found that the same printing ink chemicals may be classified differently by different suppliers. The discrepancies in chemical classification stem partially from differences in classification of chemicals across various countries, data availability, interpretation of data and different concentration cut-offs.
As such, users of the raw material are often left with the task of assessing the data and resolving the classification discrepancies.
This is a crucial issue, especially for chemicals that may be classified as CMR (carcinogens, mutagens or reproductive toxins). To provide clarity and eliminate confusion in the customer base (i.e. printers), the NAPIM SDS Evaluation Committee, composed of EHS experts from both ink manufacturers and raw materials suppliers, has undertaken a program aimed at identifying printing ink raw materials which are reported to have health hazard classifications that are inconsistent or conflict with the health hazard classifications of similar/identical substances.
Introduction
Classification of a chemical or a mixture is a scientific assessment of whether it has the potential to harm human health or the environment if exposure occurs. Chemicals are evaluated and classified so that people using them can understand any potential hazardous effects they could have on human health or the environment and protect against that harm.
It is vital that classification is based on accurate, robust and adequate data. The purpose of the Globally Harmonized System of Classification and Labeling of Chemicals (GHS) is to protect human health and the environment by having readily available information on the hazard properties of chemicals.
This permits the development of appropriate control measures that allow the production, transport, use and disposal of chemicals to be managed safely. The Safety Data Sheet (SDS) identifies the hazards of chemicals and mixtures and communicates the hazard information, as well as protective measures, on labels and SDS.
In 2015, the Occupational Safety and Health Administration (OSHA) integrated the GHS ver. 3 into the Hazard Communication Standard (HCS) 29CFR 1910.1200. This revised federal regulation was to be implemented in the US by June 1, 2015. The revised rule included new pictograms, signal word, hazard statements and precautionary statements appearing on the labels and SDS.
GHS is revised frequently. As of 2018, the most current GHS is version VII. OSHA adopted GHS ver. 3. The OSHA HazCom standard does not change automatically to when the GHS is revised. Classification of chemicals using the OSHA HazCom standard under Appendices A and B may not be consistent with the version of GHS in use in other geographic regions.
Accordingly, to provide clarity and eliminate confusion for employees and customers, the NAPIM SDS Evaluation committee has undertaken a program aimed at identifying printing ink raw materials which are known to have health hazard classifications that are inconsistent with the health hazard classification procedure specified in Appendix A of the HazCom standard. These materials along with their recommended health hazard classification are listed below. This applies to printing inks sold in the United States only.
Process
The NAPIM SDS Evaluation Committee, composed of experts within the NAPIM membership, has undertaken a program aimed at identifying printing ink materials which are known to have health hazard classifications that are inconsistent with the health hazard classification procedure specified in Appendix A of the HazCom standard. The printing ink materials with discrepancies in hazard classifications are nominated to the NAPIM SDS Evaluation committee.
This is an anonymous process. The committee reviews all available health hazard data (see Figure 1) to derive a consensus recommendation that is based on the available data. When a consensus health hazard classification is reached, the material along with its recommended health hazard classification is listed on the NAPIM website (see Figure 2). This is applicable to the printing inks sold in the US only.
Conclusion
The NAPIM SDS Committee process for resolving health hazard classification discrepancies utilizes collective knowledge and ensures transparency in resolving the health hazard classification discrepancies. The printing ink industry is committed to the safety of its employees and customers. Providing consistent and accurate information regarding potential printing ink health hazards to both employees and customers is essential to meeting this commitment.
Sneha Bhatia is a regulatory affairs specialist of global regulatory product stewardship at Sun Chemical. George Fuchs is the director of regulatory affairs and technology at NAPIM. Gregory Pace is the manager of global regulatory & product stewardship at Sun Chemical.