David Savastano, Editor04.15.21
Ensuring that medicines are not exposed to potentially damaging heat is critical. Amid the COVID-19 pandemic, the need to secure the pharma supply chain is even more important, as the Pfizer and Moderna vaccinations must be stored at sub-zero temperatures.
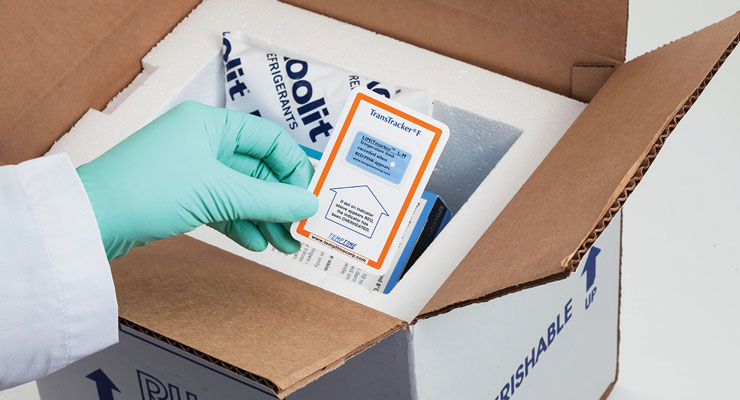
Color-changing materials presented as inks can play a significant role here. These specialized inks can show if the temperature range has been exceeded. This is an area that Temptime has been involved in for the past three decades.
Founded in 1987, Temptime was spun out of Allied Corporation. The company’s founders identified color-changing monomers that were printed as color-changing barcodes and measured with a reader that looked like a wand. This early time-temperature indicator system was used for inventory management of perishable food products. While this had proved to be a successful approach, it was ahead of its time.
“In the 1980s, you had to do everything over telephone lines, and you couldn’t transmit that amount of data,” said Chris Caulfield, VP Temptime operations, Zebra Technologies. “It took more than 30 years for the rest of the world to catch up. We evolved to a visual indicator so that people could use their eyes to look at the label and see if it had been exposed to heat.”
In 2019, Zebra Technologies acquired Temptime, which has worked out well for both companies.
“Joining Zebra has been a really good experience,” said Caulfield. “The supplies group has printing and label material expertise, and we now are part of a company that allows us to have access to handheld scanning technologies as well as cloud and data information systems. This expertise fits very well.”
Caulfield said that Temptime’s color-changing inks are the key to its technology.
“At the core of our technology, we manufacture our own color-changing inks for our heat and freeze indicators,” Caulfield said. “Our technology has been used for 30 years. The challenge in the early days was scale and capacity. All of our products are manufactured in an active state – once they are finished, they are kept in frozen or chilled environments to store them.
“At its core, our technology has not changed and that’s part of the beauty – its performance is highly projectable,” added Caulfield. “The use of our vaccine vial monitor (VVM) to facilitate the identification of potentially heat-damaged vaccine during large global vaccination campaigns is at the core of what we do. More recently, we have expanded the use of our technologies into other life science and perishable food markets. Since the acquisition by Zebra Technologies, we have expanded our vision to provide for the digitization of color-changing materials.”
The life sciences market is the most demanding on the specifications and performance of temperature monitoring technologies; outside of life sciences, the perishable food market is a potential adjacency for Temptime’s devices.
By the early 1990s, Temptime was participating in a global challenge to create a miniaturized temperature monitoring device for vaccine vials. Temptime’s researchers developed the first miniaturized technology to monitor the time and temperature exposure of vials containing the oral polio vaccine.
“That’s how we entered the vaccine space,” Caulfield noted. “We have unique technology that can identify if vaccines have been exposed to potentially damaging heat. For example, the oral polio vaccine has similar storage and shipment requirements as the Moderna COVID-19 vaccine at 20°C.
“Vial level monitoring is one of the critical elements to a successful vaccine outreach program,” Caulfield added. “There are mass vaccination programs that the rest of the world uses. For example, India mobilized volunteers to combat polio, with 100 million doses in three days. In other countries, it is standard practice to bring the vaccines to where the people are, and our technology helps facilitate that.”
Temptime is working with companies that are handling the COVID-19 vaccines and is involved in COVID-19 vaccine distribution.
“We manufacture medical devices that use our color-changing ink formulations, as well as the VVM. Each use case for our products must be evaluated to determine if there will be a regulatory requirement and potential regulatory filing,” he noted.
“In the case of the VVM, we are selling directly to the vaccine manufacturer and the technology becomes part of the labeling of the vaccine. The vaccine manufacturer works with their country’s regulating body and/or the World Health Organization to satisfy the regulatory requirements.”
Temptime Corporation
116 The American Road
Morris Plains, NJ 07950
Tel: 973-630-6000
Email: info@temptimecorp.com
www.temptimecorp.com
Specialties: Color changing inks for heat and freeze indicators.
Color-changing materials presented as inks can play a significant role here. These specialized inks can show if the temperature range has been exceeded. This is an area that Temptime has been involved in for the past three decades.
Founded in 1987, Temptime was spun out of Allied Corporation. The company’s founders identified color-changing monomers that were printed as color-changing barcodes and measured with a reader that looked like a wand. This early time-temperature indicator system was used for inventory management of perishable food products. While this had proved to be a successful approach, it was ahead of its time.
“In the 1980s, you had to do everything over telephone lines, and you couldn’t transmit that amount of data,” said Chris Caulfield, VP Temptime operations, Zebra Technologies. “It took more than 30 years for the rest of the world to catch up. We evolved to a visual indicator so that people could use their eyes to look at the label and see if it had been exposed to heat.”
In 2019, Zebra Technologies acquired Temptime, which has worked out well for both companies.
“Joining Zebra has been a really good experience,” said Caulfield. “The supplies group has printing and label material expertise, and we now are part of a company that allows us to have access to handheld scanning technologies as well as cloud and data information systems. This expertise fits very well.”
Caulfield said that Temptime’s color-changing inks are the key to its technology.
“At the core of our technology, we manufacture our own color-changing inks for our heat and freeze indicators,” Caulfield said. “Our technology has been used for 30 years. The challenge in the early days was scale and capacity. All of our products are manufactured in an active state – once they are finished, they are kept in frozen or chilled environments to store them.
“At its core, our technology has not changed and that’s part of the beauty – its performance is highly projectable,” added Caulfield. “The use of our vaccine vial monitor (VVM) to facilitate the identification of potentially heat-damaged vaccine during large global vaccination campaigns is at the core of what we do. More recently, we have expanded the use of our technologies into other life science and perishable food markets. Since the acquisition by Zebra Technologies, we have expanded our vision to provide for the digitization of color-changing materials.”
The life sciences market is the most demanding on the specifications and performance of temperature monitoring technologies; outside of life sciences, the perishable food market is a potential adjacency for Temptime’s devices.
By the early 1990s, Temptime was participating in a global challenge to create a miniaturized temperature monitoring device for vaccine vials. Temptime’s researchers developed the first miniaturized technology to monitor the time and temperature exposure of vials containing the oral polio vaccine.
“That’s how we entered the vaccine space,” Caulfield noted. “We have unique technology that can identify if vaccines have been exposed to potentially damaging heat. For example, the oral polio vaccine has similar storage and shipment requirements as the Moderna COVID-19 vaccine at 20°C.
“Vial level monitoring is one of the critical elements to a successful vaccine outreach program,” Caulfield added. “There are mass vaccination programs that the rest of the world uses. For example, India mobilized volunteers to combat polio, with 100 million doses in three days. In other countries, it is standard practice to bring the vaccines to where the people are, and our technology helps facilitate that.”
Temptime is working with companies that are handling the COVID-19 vaccines and is involved in COVID-19 vaccine distribution.
“We manufacture medical devices that use our color-changing ink formulations, as well as the VVM. Each use case for our products must be evaluated to determine if there will be a regulatory requirement and potential regulatory filing,” he noted.
“In the case of the VVM, we are selling directly to the vaccine manufacturer and the technology becomes part of the labeling of the vaccine. The vaccine manufacturer works with their country’s regulating body and/or the World Health Organization to satisfy the regulatory requirements.”
Temptime Corporation
116 The American Road
Morris Plains, NJ 07950
Tel: 973-630-6000
Email: info@temptimecorp.com
www.temptimecorp.com
Specialties: Color changing inks for heat and freeze indicators.